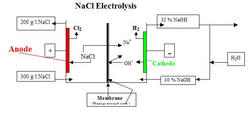
Electric current is carried through the NAFION* membrane by Na+ ions. At the cathode, water is split into OH- ions (they combine with Na+ -> NaOH) and hydrogen by the addition of electrons. At the anode, Cl- ions are discharged -> chlorine.
/images/nacl5.jpg
/images/nacl5_250x117.jpg